|
3/20/2024 0 Comments Periodic table reactive metals They get more reactive the further down on the periodic table you go too, with cesium and francium being so reactive that they can burst into flames simply by being exposed to the air. 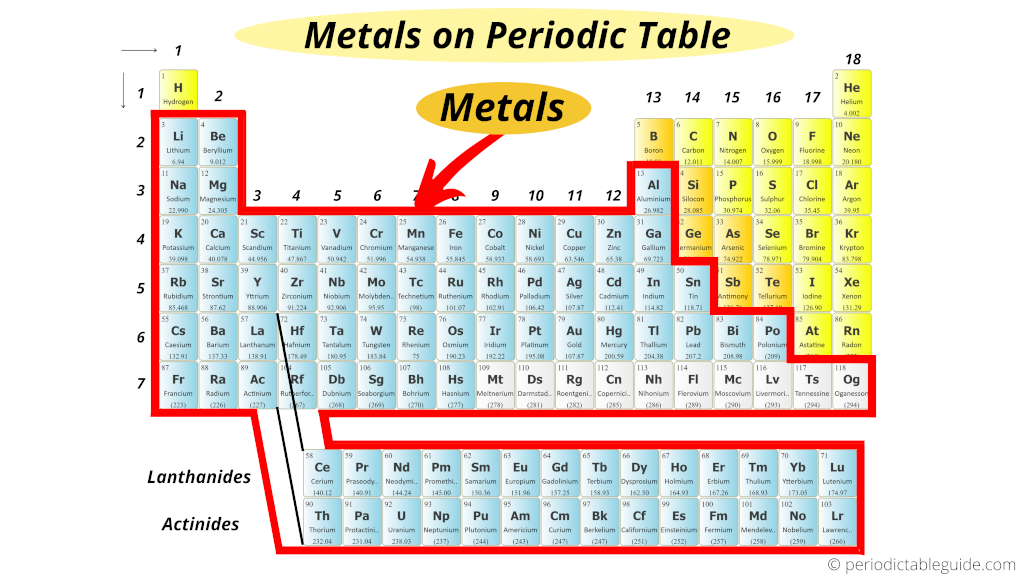
To remember how the reactivity of the alkali metals and halogens increases or decreases, put a pin in the middle of the periodic table and spin it anti-clockwise. Alkali metals react vigorously to water and air.These elements will dance around, sizzling due to the production of hydrogen gas, and often explode. The outer shell will more easily attract another electron, which needs an electron to complete its full outer shell, when there is more attractive force.Ī useful mnemonic picture to help you recall that:Īs you go up group 7 (the halogens), again the elements get more reactive. The fewer electron shells (rings) between the nucleus and the outer shell (ring) also has less shielding effect and again this increases the electron attraction. The distance "a" is less than "c" and the force of attraction between the nucleus and the outer shell increases with shorter distances. A more reactive metal donates electrons more readily to form a cation. Refresher: The periodic table is organized into groups - where each column comprises a group. They are all metals and increase in reactivity down the group and when reacted with water form alkali solutions. The reactivity series tells us how much a metal wants to be a cation. Group 1 is the first group in the periodic table containing elements that are commonly known as the Alkali metals. For example, rubidium has 5 electron shells, a property found in all other period 5 elements, whereas its electron configuration s ending is similar to all other alkali metals: s 1. More reactive metals react more vigorously, increasing the rate of effervescence of hydrogen gas. It is an alkali metal, the most reactive group in the periodic table, having properties and similarities with both other alkali metals and other period 5 elements. The most reactive metal is francium, the last alkali metal (and most expensive element).However, francium is an unstable radioactive element, only found in trace amounts. Halogens from bromide to fluorine get more reactive because the force of attraction between the nucleus (core) and the outer electron get stronger as you go up group 7 elements. Secondly, reactivity affects the rate of reaction. The most reactive element is fluorine, the first element in the halogen group. Why do halogens get more reactive going upwards in group 7? The outer electron is more easily transferred to say an oxygen atom, which needs electrons to complete its full outer shell. 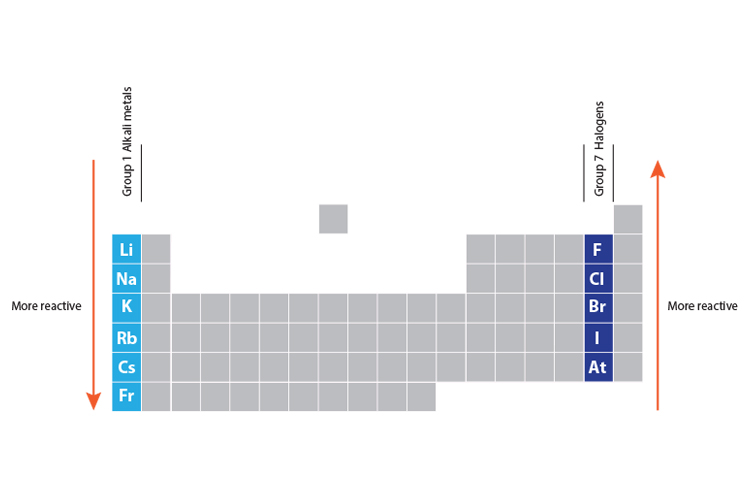
The more electron shells (rings) between the nucleus and outer electron also creates shielding and again this weakens the nuclear attraction. 
The distance "c" is greater than "a" and the force of attraction between the nucleus and the outer shell (rings) diminishes with distance. Why do alkali metals get more reactive going down group 1?Īlkali metals from lithium to potassium get more reactive because the force of attraction between the nucleus (core) and the outer electron gets weaker as you go down group 1 elements. As you go down group 1 (the alkali metals) in the periodic table, the elements get more reactive.Īs you go up group 7 (the halogens), again, the elements become more reactive. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
